Revenue.
Margin.
Valuation.
Three numbers your board reads. AI is rewriting all three in your industry.
This briefing tells you where revenue, margin, and valuation move in medtech as AI rewrites the industry. Read it before your competitors decide what your next decade looks like.
The 10-page briefing. Worth 20 minutes.
One email. One PDF. Worth twenty minutes of your week.
We send it once. Work emails only.
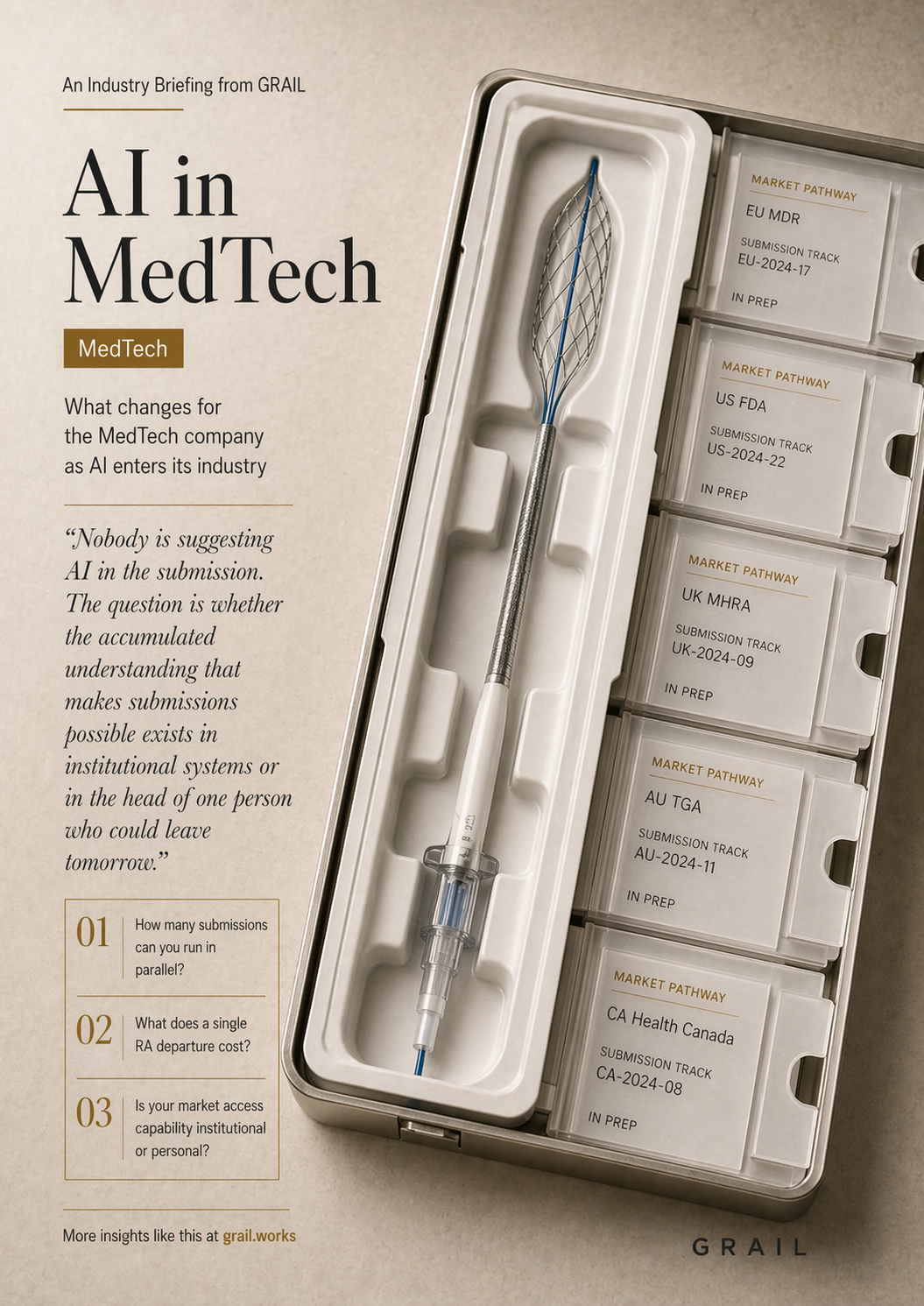
A mid-market MedTech company competes against the same regulatory complexity as organizations ten times its size. MDR compliance, notified body interactions, post-market surveillance, clinical evidence architecture. The large-cap firm spreads this knowledge across a department of forty specialists. The mid-market firm concentrates it in one or two regulatory affairs professionals whose departure would delay a CE mark submission by twelve to eighteen months.
Nobody is suggesting AI in the submission. The question is whether the accumulated understanding that makes submissions possible exists in institutional systems or in the head of one person who could leave tomorrow.
At the same time, hospital procurement across Europe is shifting from clinical champion adoption to evidence-based evaluation. EU HTA extends to high-risk devices in 2026. Commercial teams built on twenty years of physician relationships do not have health economics capability. Two fronts. Same leadership team. Same window closing.
For the CEO of a MedTech company, this is not a regulatory question. It is a revenue question, a profit question, and a valuation question.
Revenue. Profit. Valuation.
Three lenses. Three answers the management team needs before the next board meeting.
Revenue
Revenue in MedTech is gated by regulatory speed. Every month a CE mark is delayed is a month of lost market access. When regulatory capacity is concentrated in one or two people, the number of parallel submissions is capped by their bandwidth. A competitor who builds institutional regulatory infrastructure runs three submissions while you run one. The revenue gap compounds with every quarter.
Profit
A senior regulatory affairs director's departure delays submissions by twelve to eighteen months. At typical device revenue trajectories, that delay costs millions in market timing. The retention cost of a single specialist is a fraction of the revenue at risk. Knowledge infrastructure that eliminates this single-point-of-failure is insurance with immediate ROI.
Valuation
An acquirer or investor evaluating a MedTech company prices regulatory capability directly. Personal expertise in two heads is a risk discount. Institutional knowledge infrastructure encoded into precedent databases, structured submission processes, and evidence systems earns a premium. Same pipeline, same revenue. Different multiples depending on whether the capability survives a leadership transition.
What you'll get when you download
A 10-page report for European mid-market MedTech CEOs. Designed to be read in one sitting before your next board meeting.
The strategic choice, side by side
The default path (rely on individual experts, react to evidence demands, cap submissions at specialist bandwidth) and the repositioning path (build knowledge infrastructure, scale regulatory throughput, earn the institutional premium), with the financial logic of each.
The four levers that compound
Encode regulatory knowledge before specialists leave. Build health economics evidence infrastructure before procurement demands it. Free specialist capacity from documentation to judgment. Convert submission history into a compounding precedent asset.
Five questions for your next board meeting
Diagnostic questions the CEO should test the leadership team against before the board revisits key-person risk and market access strategy. The questions where the room cannot agree are the ones worth a longer conversation.
Calibrated for each seat at the table.